

Dissolved oxygen sensors in a nutshell
All industries however related to water, e.g. heating or aquaculture, measure the oxygen dissolved in water. In the following article, we answer questions related to this process - you will find out, among other things, what types of oxygen sensors there are, what their advantages and disadvantages are and how the measurement takes place.
Where is dissolved oxygen measured in water?
In all water-related industries, the dissolved oxygen content of the water is measured. This parameter is important for the functioning of aquatic ecosystems - it reflects their 'health', i.e., among other things, the balance between oxygen supply from the atmosphere and photosynthesis. It also provides information on water quality.
Oxygen control is therefore necessary, among other things, in heating systems, where the presence of oxygen is a sign of corrosion, as well as in fish farms, where it is essential for the survival of fish and plants.
What is an acceptable level of dissolved oxygen?
Healthy water should have a general dissolved oxygen concentration above 6.5-8 mg/L and between about 80-120%.
Oxygen meters – applications
Typical applications where oxygen sensors are used:
-
Drinking water monitoring
-
Fish farming (fresh and salt water)
-
In municipal and industrial wastewater treatment plants
-
General water and wastewater technology
-
Universities and educational institutions

Dissolved oxygen sensors are used, among other things, in fish farming
Basis for measuring dissolved oxygen concentration in water
Unit of measurement – dissolved oxygen value
Oxygen is measured, for example, in mg/l, ppm, ppb or %/volume.
Influence on measurement values
It should be taken into account that the measurement result is influenced by values such as:
-
temperature of the measuring solution - this can be compensated for by an integrated temperature sensor as in the JUMO digiLine O-DO S10 oxygen probe
-
ambient air pressure
-
salinity
Electrometric versus optical sensors ro measure dissolved oxygen in water
There are two types of sensors for measuring dissolved oxygen in water: electrometric and optical.
Electrometric dissolved oxygen sensors
Electrometric sensors, measure the concentration of dissolved oxygen in water based on the electrical current generated.
Using the electrometric method of measuring oxygen, decomposition of the anode and wear and tear of the electrolyte during operation cannot be avoided.
Optical dissolved oxygen sensors
Optical sensors, also known as luminescence sensors, measure the concentration of dissolved oxygen in water based on the extinction of luminescence in the presence of oxygen by oxygen permeable membrane.
Luminescence – the principle of optical oxygen sensors
Luminescence is a highly sensitive test method that helps to detect various substances. In optical oxygen sensors, measurement using it proceeds as follows:
1. The sensor has a sensor cap with an internal luminophore coating.
2. A light source inside the sensor cyclically illuminates the luminophore.
3. The phosphor, by absorbing energy, changes its ground state to an excited state. After some time, it returns to its ground state, losing heat and emits the remaining energy in the form of red light (called fluorescent radiation), which is detected by a photodiode in the sensor body.
4. In the presence of oxygen, the phosphor collides with it and the emission of luminescent radiation is not stopped.
5. No oxygen is consumed in the process and no minimum flow rate is required. This measurement method allows high measurement accuracy even at low oxygen concentrations.
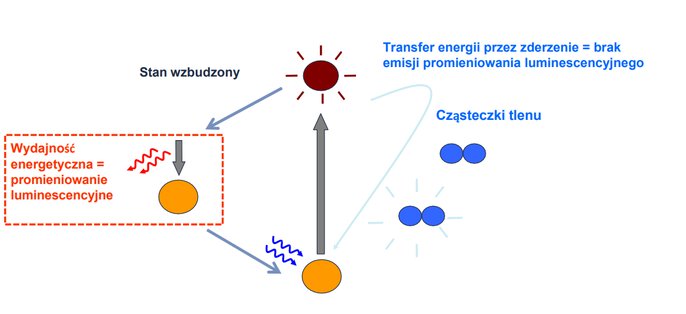
Optical oxygen sensor – operating principle
Optical dissolved oxygen meter for water and wastewater JUMO digiLine O-DO S10
An example of an optical oxygen sensor for measuring the content of dissolved oxygen in water and other liquids is the digital probe JUMO digiLine O-DO S10. Thanks to its precision in a wide measurement range, it is used in various industrial applications, such as fish farming, wastewater treatment plants, surface water, or laboratory applications. One of the main advantages of this device is the lack of the need to replenish electrolytes and the infrequent need for calibration. The probe is equipped with both digital and analog interfaces, allowing versatile application in different measuring systems. Additionally, the sensor's self-diagnostic functions provide constant monitoring of its condition and rapid detection of any disturbances. The measured value for dissolved oxygen is automatically compensated for temperature. Additionally, using external sensors, the measured value can be compensated for air pressure and salinity of the medium.
We answer some of the most frequently asked questions about our oxygen meter:
Is the JUMO digiLine 0-D0 S10 oxygen probe suitable for installation in untreated wastewater?
What are the measurement values in mg/l for the digiLine 0-D0 S10 oxygen probe?
The JUMO digiLine O-DO S10 sensor is used to measure the concentration of oxygen in the range of 0 to 20 ppm (mg/l) (or 0 to 200% saturation).
What modes of operation does the digiLine 0-D0 S10 oxygen probe have?
The JUMO digiLine O-DO S10 sensor has the following modes of operation:
- Communication via digital interface through RS485 Modbus RTU protocol.
- Transmission of analog signal via a two-wire current interface of 4 to 20mA.
Optical dissolved oxygen sensor – advantages
The advantages of luminescence-based optical dissolved oxygen measuring instrument, compared to electrometric sensor, are:
-
stability and precision of measurement over a longer period of time - calibration is carried out infrequently
-
less maintenance work
-
no change of electrolytes
-
no polarisation voltage required
-
no minimum inflow
-
low drift
- ${title}${badge}

