

The pH electrode - everything you need to know
The pH value is one of the most commonly measured parameters in liquid analysis. To determine it, pH electrodes are commonly used. These make it possible to measure acidity and alkalinity continuously throughout the process. Check out our FAQ - it will give you the answers to the most frequently asked questions about pH electrodes and help you choose the right instrument for your application.
What is a pH electrode?
A pH probe is used to measure the pH value using a glass membrane that is sensitive to H+ ions. The device measures their activity in a solution, from which its alkalinity or acidity can be determined.
What is a pH electrode made of?
The pH electrode consists of two electrochemical half-cells: indicator and comparison. These are connected by a sensitive voltmeter, which converts the values of the electromotive force, the so-called SEM of the solutions, into a result from the pH scale. A glass membrane, which has a negative charge, forms the measuring system.
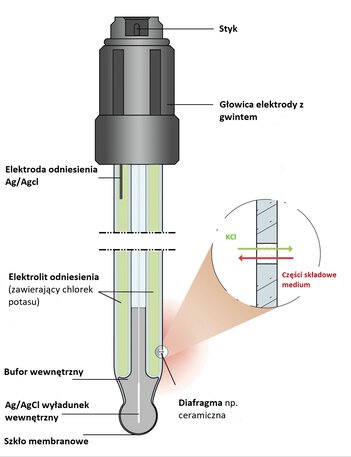
pH electrode construction
How does the pH electrode work?
The pH electrode measures the potential difference between the reference electrode and the glass pH electrode. This difference depends on the concentration of H+ ions in the solution, i.e. the acidity/pH of the solution.
Slope of pH electrode - characteristics
The slope of the pH electrode characteristic shows how the potential of a given pH probe changes under the influence of a tenfold change in the concentration of the ion to which the electrode is sensitive. In the case of a glass electrode, this is the change in its potential per pH unit [mV/pH].
Which electrodes for pH measurement?
The pH is measured using different types of electrodes:
-
glass electrodes (measuring electrodes) with a pH-sensitive glass membrane and a reference electrode with a potential as independent as possible of pH and temperature
-
Combination electrodes - glass electrode and reference electrode combined in one unit. Combination electrodes work by having a sensing half-cell (AgCl covered silver wire) and a reference half-cell (Ag/AgCl reference electrode wire). These two components must be joined together to complete a circuit in order for the ph meters to get a pH reading.
Today, combination pH electrodes, known as combination electrodes, are most commonly used for pH measurement. They are much more convenient in measurement practice.
Glass body electrodes and epoxy body electrodes
Due to their mechanical resistance, epoxy body electrodes are suitable for demanding applications, e.g. and their disadvantage is a lower thermal resistance compared to glass electrodes. They can now also be used to determine the pH of solutions containing organic solvents and corrosives. In contrast, glass electrodes can withstand higher operating temperatures and highly corrosive solvents.
What is the difference between a single junction and double junction electrode?
Double junction electrodes have an additional salt bridge to prevent reactions between the electrode filling solution and the sample. These reactions could damage the electrode junction. Double junction electrodes are used to test samples that contain proteins, heavy metals or sulphides.
Single junction electrodes - for general appliactions
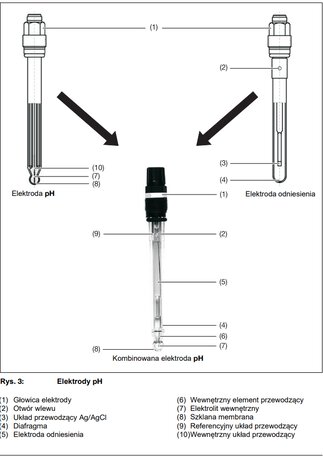
Combined pH electrode
Why is a reference electrode needed when measuring pH with a laboratory pH electrode?
A reference electrode - made of glass insensitive to H+ ions and open to the sample environment - is essential to ensure a defined and stable reference potential. This is important because the potential of the pH electrode will be measured against it. Both electrodes must be immersed in the same solution.
How to clean the pH electrodes?
The pH probe should be cleaned regularly so that no material builds up on the surface of its glass bulb. Otherwise the electrode will calibrate, making its readings inaccurate.
Tips for effective pH electrode cleaning
In most cases, warm water with dishwashing liquid is sufficient to remove grease and oil. Lime or iron oxide deposits can be removed with vinegar, citric acid or dilute hydrochloric acid. Never clean the membrane mechanically. Even wiping or drying it can lead to errors in the measuring function. After cleaning, rinse the electrode with deionised (distilled) water.
How often should I clean the electrode?
The minimum frequency of electrode cleaning is once a month. In addition, this activity is also necessary before each calibration.
How to store pH electrodes when not in use?
Storing a pH electrode in a few steps:
-
Close the electrolyte top-up hole, if present.
-
Fill the protective cap with electrolyte solution.
-
Insert the cleaned electrode into the protective cap.
-
During prolonged storage, check regularly that there is sufficient electrolyte in the protective cap.
-
Store the electrode in such a way that moisture does not get into the connector.
Never store pH electrodes in deionised water as it depletes the ion-rich reference electrolyte. In some cases it is possible to use pH 4 or 7 buffers instead of the solution.
How often should I change the storage solution for pH electrodes?
If the electrode is stored for more than six months, the KCl solution in which it is immersed should be replaced after this time.
How to calibrate a pH electrode?
After starting the measurement, the transmitter must be matched to the electrode. Three methods are available for this purpose: one-point, two-point and three-point calibration.
-
Single point calibration is the optimum method for applications where a comparative measurement can only be performed with a hand-held instrument. In this method, the pH value is measured as close as possible to the measuring point of the transmitter, using a calibrated handheld meter. The value indicated by the transmitter is then set to the value of the handheld meter by adjusting the system zero point.
-
Two-point calibration is the most commonly used method for measuring pH. Two buffer solutions are used for calibration, e.g. pH = 7 and pH = 4. Although pH transmitters allow any order of buffer solutions, it makes sense to start with the neutral solution pH = 7.
-
Three-point calibration - we recommend this if the calibration needs to cover a particularly wide range, e.g. pH = 4 to pH = 9.
Step-by-step calibration of the pH measuring electrode
-
Rinse the electrode in the rinsing solution with a vigorous movement.
-
Shake the electrode to remove any residual solution.
-
Dip in standard solution.
-
Wait for the reading to stabilise.
-
Take a reading and record the known pH value of the standard solution.
-
Repeat for any number of points.
How often calibrate the pH meter?
The PH meter should be calibrated even daily, always before a series of measurements. Calibration of the pH electrode should also be carried out after each maintenance operation (e.g. cleaning or replacement of the electrode), at the latest after about 3 to 4 weeks of operation.
JUMO calibration buffers
How to choose a pH sensor? Criteria
How to correctly select a pH electrode for a specific application? This question is very important because the choice of electrode affects its service life. To choose the right sensor, you should:
-
Check the parameters of the medium and installation in which the electrode is to operate, such as the type of measuring medium, temperature and pressure, chemical composition or pH range, among others.
-
Select a suitable diaphragm. The potential of the reference electrode is more stable the more permeable the diaphragm is.
-
Decide whether the electrode should include an integrated temperature sensor, such as the JUMO tecLine pH.
-
Taking into account the type of application, select the type of membrane glass from which the measuring part of the electrode is made.
Does JUMO have pH electrodes for the food industry? ISFET pH combination electrode
The ISFET pH combination electrode, due to its robust construction and resistance to mechanical damage, fast response time and high pH measurement accuracy even at low temperatures, can be successfully used in places such as:
-
food and beverage establishments
-
dairies, cheese factories
-
all hygienic production processes
-
production processes with 3-A requirements
The sensor fulfils the hygienic requirements according to 3-A.

ISFET pH combination electrode for industries such as food and beverage
PH electrode - when does it need to be replaced? Service life
pH electrodes are subject to wear and must be replaced regularly. The degree of wear of the electrode can be recognised by the appearance of the salt rings, as they are washed away over time and disappear. The life of a pH electrode is limited due to the gradual ageing of the sensor and lasts for approximately 360 readings (or one year with one reading per day). It is virtually impossible to determine the exact lifetime as it is affected by many factors.
How do I care for my pH meter?
A few key rules to follow to enjoy the longest possible electrode life:
-
Suitable position, vertical between 0 and 35°. The electrical connection should be 10° higher than the measuring diaphragm.
-
Periodic calibration
-
Storage in containers with liquid KCl to avoid drying out the pH electrode.
-
Regular cleaning, especially when probes work in contaminated media.
Which pH probe for an aquarium? JUMO ecoLine/ JUMO BlackLine pH
The combination probes of the JUMO ecoLine / JUMO BlackLine pH series can be used to measure the pH of water in aquaria, including marine aquaria. The operating temperature of these electrodes is between 0 and 60°. Furthermore, these devices are characterised by their excellent value for their price.

Combined pH electrode JUMO ecoLine / JUMO BlackLine pH
What are state-of-the-art pH electrodes for demanding applications? JUMO tecLINE HD
JUMO tecLINE HD pH sensors are designed for the most demanding applications, for process and industrial applications. They also include a built-in pt1000 temperature sensor.
Typical areas of application:
-
chemical industry
-
process water
-
industrial waste water engineering: process waste water, waste water with highly contaminated media: presence of oil or electrode poisons
-
galvanic: process effluents, media containing electrode poisons (metal ions, complexing agents)
-
power plants and waste incineration plants: flue-gas cleaning
-
sugar industry: constant high temperature, presence of electrode poisons (e.g. sulphides).

pH combination electrodes JUMO tecLine HD
What temperature and pressure can pH probes withstand?
Our pH electrodes, which can be used on pipelines, for example, have different ranges of permissible temperature and pressure depending on the model. For example, JUMO tecLine HD models can operate at temperatures from 0 to 135 °C and withstand process pressures of up to 13 bar. Other models, such as the JUMO ecoLine/JUMO BlackLine pH, are suitable for temperatures from 0 to 60 °C and pressures from 0 to 6 bar. More information on specific models can be found on our website.
- ${title}${badge}






